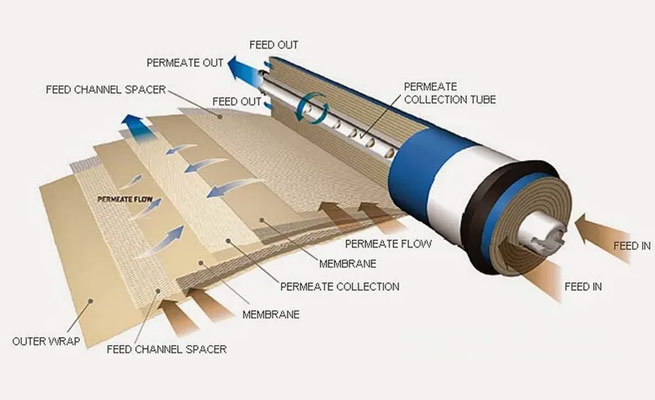
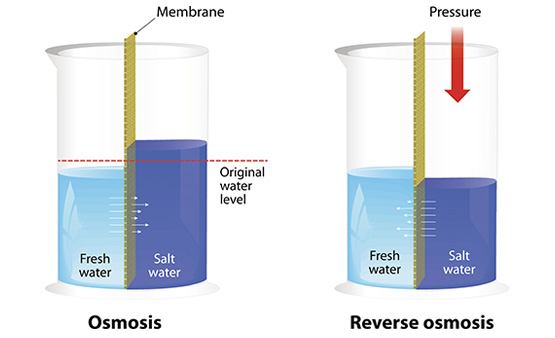
Overview
Reverse osmosis (RO) is a water purification technology that uses a semipermeable membrane to remove ions, molecules, and larger particles from drinking water. In RO water purifiers, an applied pressure is used to overcome osmotic pressure, a colligative property, that is driven by chemical potential differences of the solvent, a thermodynamic parameter. Reverse osmosis can remove many types of dissolved and suspended species from water, including bacteria, and is used in both industrial processes and the production of potable water. The result is that the solute is retained on the pressurized side of the membrane and the pure solvent is allowed to pass to the other side. To be "selective", this membrane should not allow large molecules or ions through the pores (holes), but should allow smaller components of the solution (such as solvent molecules) to pass freely.
RO water purifiers also involves diffusion, making the process dependent on pressure, flow rate, and other conditions. RO water purifiers is most commonly known for its use in drinking water purification from seawater, removing the salt and other effluent materials from the water molecules.
Please leave a message for more details and we will get back to you soon.
Enquire Now
- Customer Care Services
+1800-103-1145
- Talk to Us